- Blog
- Charles barkley daughter charles barkley family
- Download microsoft office 2019 full crack
- Best video editing apps for youtube windows
- Padlock with code
- Counter strike source texture pack gmod
- 1960 dallas cowboys roster
- Printable simple monthly expense tracker
- Siberian mouse adult babko naked
- Asd dsm 5 APA
- Emcee send off wedding script
- Fs17 straw harvest free download
- Unity funeral home deland florida
- Crazytalk animator insert sprite in composer
- Ds 160 printable form pdf
- Periodic tabl with molar mass
- Gpu cpu temp monitor
- Tabby cat personality orange
- Wacom intuos 4 installation software no disc
- Live desktop wallpaper windows 10
- Dmv duplicate title online
- Facerig live2d template
- Long bodied cellar spider reproduction

Interested in learning more about the possibilities of hydrogen education? Discover how the Horizon Hydrogen Grand Prix (H2GP) program can benefit students in a classroom near you. The result is for every 1 unit of hydrogen produced, 8 units of oxygen are created. Why does a hydrogen generator (electrolyzer) actually produce 8 times more oxygen than hydrogen? The molar mass of 2 molecules of water (2 H 20) is 18, and once the hydrogen generator splits the water into hydrogen and oxygen, the hydrogen (having a low atomic mass) only has a mass of 2 (H 2), while the oxygen has a mass of 16 (O 2).

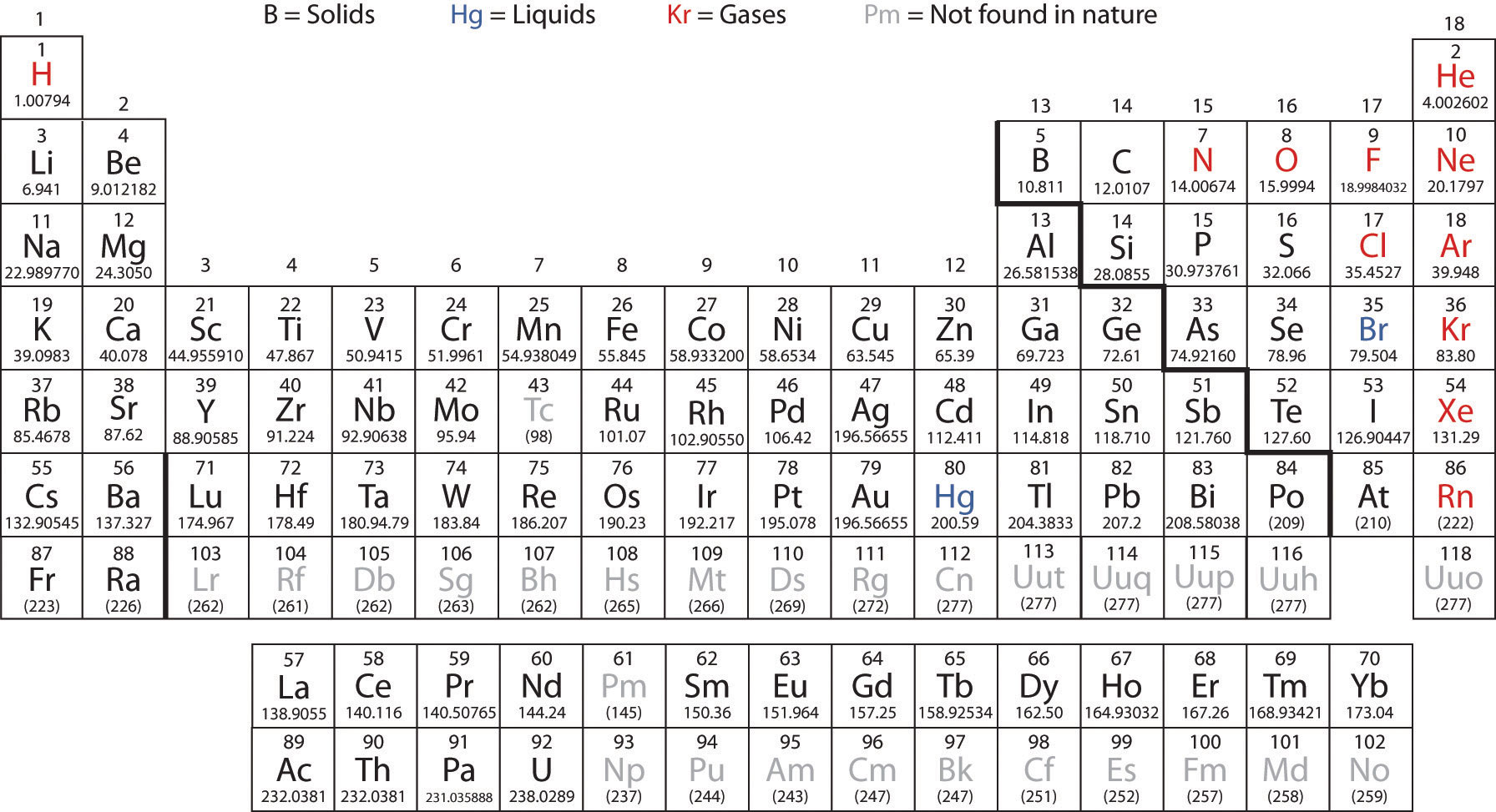
Students can understand, for instance, the process of hydrogen generation (electrolysis) with greater insight. Knowing the molar mass of hydrogen will open up a world of opportunity in terms of hydrogen education. Because of this apparent inertness, the H 2 molecule can be considered a very stable molecule at room temperature. Molecular hydrogen (H 2) can also react with many elements and compounds, but at room temperature, the reaction takes place at such a slow rate it's almost negligible. This is how hydrogen molecules – H 2 – are formed. Because of this reactivity, atoms of atomic hydrogen (H) combine with each other to form molecules of two atoms (H 2). There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. This means that overall hydrogen is an extremely ‘light’ element. The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The more protons an atom has in its nucleus, the higher the atomic number, and the element also becomes heavier. As hydrogen gas is formed of two hydrogen (H) molecules bound together (H 2) – the molar mass can be calculated as 1.00784 x 2 = 2.01568 grams.Īs hydrogen consists of only one proton, its atomic number is 1. It can be calculated by multiplying the atomic weight of hydrogen (1.00784 atomic mass units) by 2. The molar mass of hydrogen is 2.01568 g/mole. This means you can simply multiply this by 4 moles, and you’ll find out how much carbon you’ll need to weigh. You already know carbon has a molar mass of 12.01 g/mol. Take this example: say you need 4 moles of pure carbon for an experiment. This means whether you’re trying to figure out how much carbon, titanium, or cobalt you need for an experiment – understanding the molar mass is essential. Note: Although Na 2CO 3 is an ionic compound and sodium is present in an ionic form, it is a habitual way of referring to an element in these types of calculations.Molar Mass is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of a compound. This is the accurate way of rounding off to the correct number of significant figures in multistep calculations. I do it here mainly to save space, but you can keep the numbers in the calculator and round them off at the end. The little inconsistency in the final answer is due to rounding off the intermediate numbers. So, to calculate the moles from a given mass, we make a conversion factor correlating 1 mole with the molar of the given component.įor example, how many moles of sulfur are there in a 16.2 g sample? The molar mass, on the other hand, is a constant number for a given atom or a molecule as it is for a specific amount of it. The given mass is the mass of the sample, and it can be any number, for example, we can have 10 g of salt, 15 g, or 100 g. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. A modern version is shown in Figure 2.7.1 2.7. To calculate the moles from a given mass (m), the molar mass of the component is used. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. These tables are based on the 2021 table with changes from the 2019 table for the values of Ar, Hf, Ir, Pb and Yb and changes to the uncertainty for Al, Au, Co, F, Ho, Mn, Nb, Pa, Pr, Rh, Sc, Tb, Tm, and Y. The molecular mass of water is 18.0 amu, and the molar mass is 18.0 g/mol.įor the molar mass, keeping one decimal is usually an acceptable approximation, and for the Avogadro’s number, you can use 6.02 x 10 23. Numerically, the molar mass is equal to the atomic mass of a given atom or a molecule, so we can look up the molar mass of an element in the periodic table.įor example, we can say that the mass of Cu is 63.55 amu or 63.55 g/mol. The mass of one mole of atoms/molecules/ions is called its molar mass (M) expressed in g/mol. One mole of any element contains 6.022 x 10 23 of its atoms, and a mole of a compound contains 6.022 x 10 23 molecules, ions-ions, and in general, 1 mol = 6.022 x 10 23 particles. This is called Avogadro’s number ( N A = 6.022 x 10 23). By definition, a mole is the number of atoms in a 12-g sample of 12C isotope which happens to be 6.022 x 10 23 atoms.
The mole is just a number like a dozen, a hundred, or a million.
